Validation strategy

As any other medical diagnostic device, Rilemo's apparatus must be validated through a sequence of pre-clinical investigations that culminates in a first-in-human clinical study. Rilemo's pre-clinical validation portfolio is built around four complementary strands, each addressing a specific aspect of the device's readiness for the clinical phase.
Test | Year | What it validates |
In-silico validation on a 30-head digital twin | Q1 2026 | The full imaging pipeline, on the target organ (the head), on the target pathologies (haemorrhagic and ischaemic events) |
Anthropometric applicability (separate engineering strand) | Q1 2026 | The mechanical fit of the Head Unit across the full target population, validated across 18,431 Monte Carlo simulated heads |
Ex-vivo human cadaver imaging | May 2025 | The device on real human tissues, in a final clinical environment, with operative-threshold haemorrhagic targets |
Quantitative ex-vivo bovine bone imaging with CT ground truth | March 2025 | µL-scale fluid detection against CT, statistically significant on a sample large enough for analysis |
Ex-vivo bovine bone imaging | October 2024 | Fluid detection and quantification on biological samples closely resembling human tissues, against MRI ground truth |
In-vitro phantom imaging | May 2024 | The feasibility of portable fluid imaging and the usability of the user interface, against X-ray ground truth |

On in-vivo animal trials. Studies on live animals are not required for Rilemo's medical CE certification and are not planned. This was verified with four independent CROs and Notified Bodies, and is consistent with the regulatory pathway for the device class. The validation strategy proceeds directly from the strands described above to the first-in-human clinical investigation funded by this Seed round.
In-silico validation on a 30-head digital twin (Q1 2026)
This is the latest validation in Rilemo's portfolio addressing the target organ (the head) and the target pathology (intracranial haemorrhagic events).
Methodology in brief:
- The device's electromagnetic acquisition chain is simulated end-to-end in software (the "digital twin").
- Simulated acquisitions are reconstructed by the same AI pipeline that runs on the production device, producing the three-modality output described in Technology Overview.
- The dataset is built from 30 anatomically realistic head models from the IT’IS Foundation Population Head Models (PHM) repository, with tissue dielectric properties from the IT'IS Foundation database, the de facto standard for electromagnetic medical-device simulation.
- Haemorrhagic and ischaemic lesion volumes and geometries are derived from published clinical literature so that the simulated cases are coherent with realistic scenarios.
The healthy reference case is presented in the Technology Overview section. This section focuses on diagnostic discrimination between healthy and pathological cases.
Case 1: Central haemorrhagic event

- In the Tomography panel, the central haemorrhagic event introduces a localised intensity change in the affected region of the brain parenchyma, distinct from the homogeneous distribution observed in the healthy reference.
- The Radar panel highlights the boundary of the lesion as a contrast transition, isolating the haemorrhagic zone from the surrounding tissue.
- The Dielectric Permittivity panel quantifies the dielectric change in the affected region, directly correlating the imaging output with the underlying biophysical change in tissue water content that defines a haemorrhagic event.
Case 2: Wedge ischemic stroke

- In the Tomography panel, the wedge geometry of the lesion is visible as an asymmetric intensity change, geometrically distinct from both the healthy reference and the central haemorrhagic case.
- The Radar panel resolves the boundary of the wedge, supporting clinical readers in identifying the spatial pattern of the lesion.
- The Dielectric Permittivity panel quantifies the dielectric change along the wedge profile.

What this validation proves
The production AI pipeline produces clinically interpretable images of the head, healthy and pathological cases are visually and quantitatively distinguishable, and distinct lesion geometries are reproduced with the spatial characteristics expected from their underlying anatomy. This confirms that the AI pipeline reaches diagnostically meaningful image quality before the clinical investigation begins. The same pipeline is used in the cadaver and clinical phases.
Anthropometric applicability: 18,431-head Monte Carlo (Q1 2026)
In parallel with the imaging validation, Rilemo conducted a dedicated campaign to verify that the Head Unit fits the full target patient population. This work is presented separately from the imaging validation because it addresses a different question: not “does the device produce good images”, but “does the device physically work on the heads it will be used on”.
Methodology in brief:
- 18,431 anatomically realistic head models were generated through a Monte Carlo simulation drawing from multiple anthropometric data sources.
- The models span the full target patient population from 5th to 95th percentile, across all four cephalic-index categories (dolichocephalic, mesocephalic, brachycephalic, and hyper-brachycephalic).
- For each head, the placement of the 16 Head Unit antenna sections was simulated, the contact between each section and the scalp was computed, and the resulting fit was validated against design constraints.

What this validation proves
The Head Unit accommodates the full anthropometric variability of the target patient population, from the smallest 5th-percentile head to the largest 95th-percentile head, across every common skull shape category, without compromising contact quality or signal integrity. This eliminates one of the most common late-stage failure modes for head-worn medical devices.
Ex-vivo human cadaver imaging (May 2025)
The cadaver campaign was conducted at the Trecchi Human Laboratory in Cremona, Italy, with direct involvement from neurosurgeons and clinical staff. It is the first validation of the device's microwave imaging on intact human cadaveric heads, the closest pre-clinical proxy to the first-in-human investigation.
A peer-reviewed manuscript, "Detecting Intracranial Hemorrhage via Wearable Microwave Imaging: Ex Vivo Human Study", by Giaccaglia, Conti, Lidoni, Rosa, Marandici, Capitanio, Donofrio, and Fioravanti, is currently under peer review.
Methodology in brief
Three intact adult human cadaver heads were sourced from the Trecchi Human Laboratory. For each, a craniotomy was performed by neurosurgical co-authors from IRCCS Ospedale San Raffaele (Milan) and ASST Cremona, and a catheter was inserted and inflated with a blood-mimicking fluid. Two target volumes were tested per specimen, 15 mL and 30 mL, corresponding to the operative intervention thresholds at which international neurosurgical guidelines require the surgeon to act. Six experimental scenarios were obtained in total.
Each scenario followed a three-step protocol: baseline scan, controlled insertion of the simulated bleed, and second scan immediately after, without repositioning the device, allowing isolation of the bleed signal. Optical ground truth was used to measure localisation accuracy. Temperatures were continuously monitored. Image reconstruction used a beamforming algorithm with clutter-suppression pipeline.
Results

Across the six experimental scenarios, the device reconstructed the location of the intracranial bleed with a mean localisation error of 14.7 mm, with four of the six scenarios localising the target within 20mm of the optically-determined ground truth, and precision down to 1.7 mm on the best-performing acquisitions, on both the 15 mL and 30 mL target volumes. A clear haemorrhagic detection was achieved across all six scenarios, including those on the third specimen, where the larger localisation errors observed are still smaller than the anatomical regions used in clinical triage protocols and therefore remain clinically meaningful for the triage decision the device is designed to support.
The findings from all six scenarios, including the limitations encountered on the third specimen, directly informed the design of the next-generation reconstruction pipeline currently in testing, which uses a deep-learning, non-differential approach that does not require a baseline scan and is being trained on a much larger in-silico dataset.


What this validation proves
Wearable microwave imaging detects intracranial haemorrhagic targets at clinically relevant volumes, on intact human cadaveric heads, in a final clinical environment, with practising neurosurgeons involved. Detection was achieved across all six scenarios with localisation precision sufficient for triage decisions. To the authors' knowledge, this is one of the first experimental validations of a wearable microwave imaging system on intact human cadavers.
The cadaver work was performed on a research prototype. The industrialised architecture frozen in Q1 2026 incorporates the engineering improvements directly informed by these experiments. A second cadaver campaign on the industrialised prototype is planned during 2026 as part of the pre-clinical validation.
Quantitative ex-vivo bovine bone imaging with CT ground truth (March 2025)
In March 2025, Rilemo conducted a quantitative ex-vivo bovine bone imaging campaign in collaboration with the IRCCS Sant'Ambrogio Galeazzi Hospital (Milan), expanding the October 2024 work (below) along three dimensions:
- a larger sample size,
- a more rigorous ground truth (clinical CT instead of MRI)
- the detection of fluid inclusions at the µL scale.
The full study is reported in the paper "Bone-marrow oedema estimation with a microwave imaging OECP array in ex-vivo bovine long bones: comparison with CT ground truth", by Conti, Giaccaglia, Lidoni, Rosa, and Prof. Luca Maria Sconfienza (Head of Diagnostic and Interventional Radiology at IRCCS Galeazzi-Sant'Ambrogio and Full Professor at the University of Milan. The manuscript has been peer-reviewed, approved, and is currently awaiting publication.
Methodology in brief
Seven bovine bone samples (the distal and proximal halves of two femurs and two humeri from a single young animal) were prepared with a regular matrix of small drilled holes (16 to 25 holes per sample, approximately 25 µL of fluid per hole). Each hole was progressively injected with an iodine-enhanced gel via calibrated syringe, with the actual injected volume independently verified by precision weight balance.
Each sample was imaged before and after injection by a microwave imaging probe array, and the same samples were independently scanned on a clinical CT scanner at the IRCCS Galeazzi radiology department. The CT measurements provided an independent quantitative ground truth at the µL scale, against which the microwave-derived fluid volumes could be statistically compared.
Results

Across the seven samples, the microwave-derived fluid volume estimates correlated significantly with the directly measured injected volumes (Pearson r = 0.78, p = 0.037), with a small positive bias of approximately +10 %. Agreement with the CT-derived volumes was also positive but weaker (r = 0.58), as the CT comparison used a deliberately conservative threshold for marking each drilled hole as filled.


What this validation proves
The device detects and quantifies fluid inclusions in biological tissue at the µL scale, with results correlating significantly with both directly measured volumes and CT ground truth. This is Rilemo's first peer-reviewed validation against a clinical CT scanner, co-authored at the institutional level with the Head of Radiology at IRCCS Galeazzi-Sant'Ambrogio. Although this work uses a different anatomical target, it validates the underlying detection principle central to the device's imaging approach.
Ex-vivo bovine bone imaging (October 2024)
The ex-vivo bovine bone campaign was conducted in a private hospital radiology department with direct involvement and feedback from the head of Radiology, technicians, and clinical staff. The primary goal was to demonstrate the device’s ability to generate reliable images on biological samples closely resembling human tissues, validated against MRI as ground truth. The secondary goal was to validate the user experience of the device in a clinical radiology environment.
Methodology in brief
A set of femur and humerus bones from a young bovine (under 14 months) were imaged. Edema-simulating fluid mixed with a gadobutrol-based MRI contrast agent was injected into the spongy bone of each sample. The contrast agent was used solely to enable the MRI to visualise the injected fluid; it played no role in Rilemo’s measurement. Acquisitions were taken before and after injection, with 5 mL of fluid per sample, and the resulting datasets were compared in their fluid detection and quantification ability.
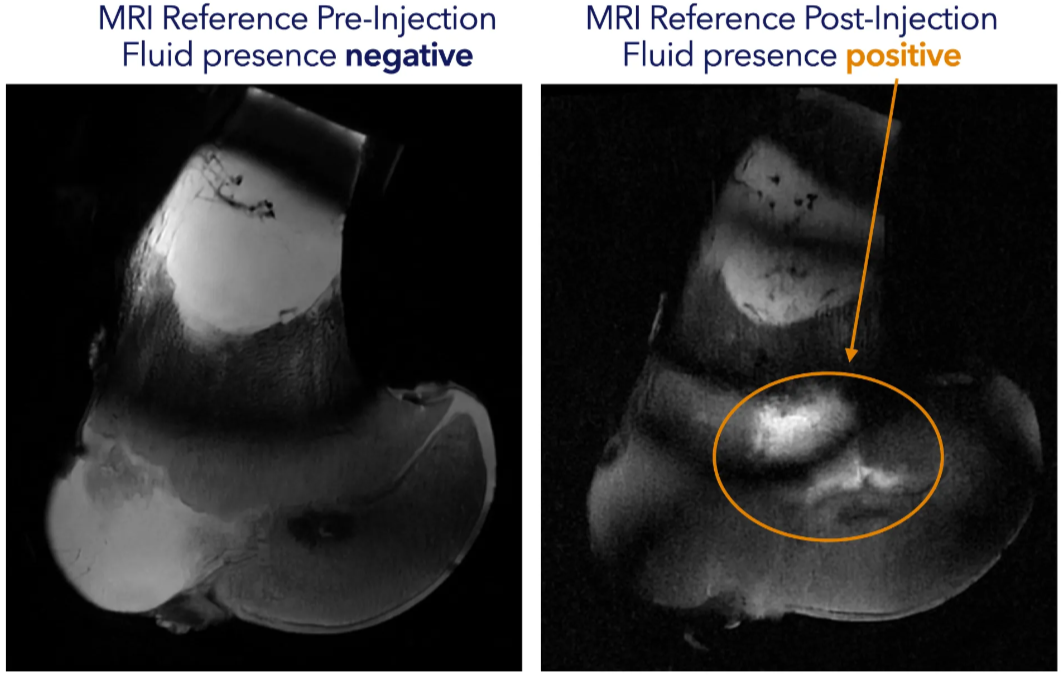
Results
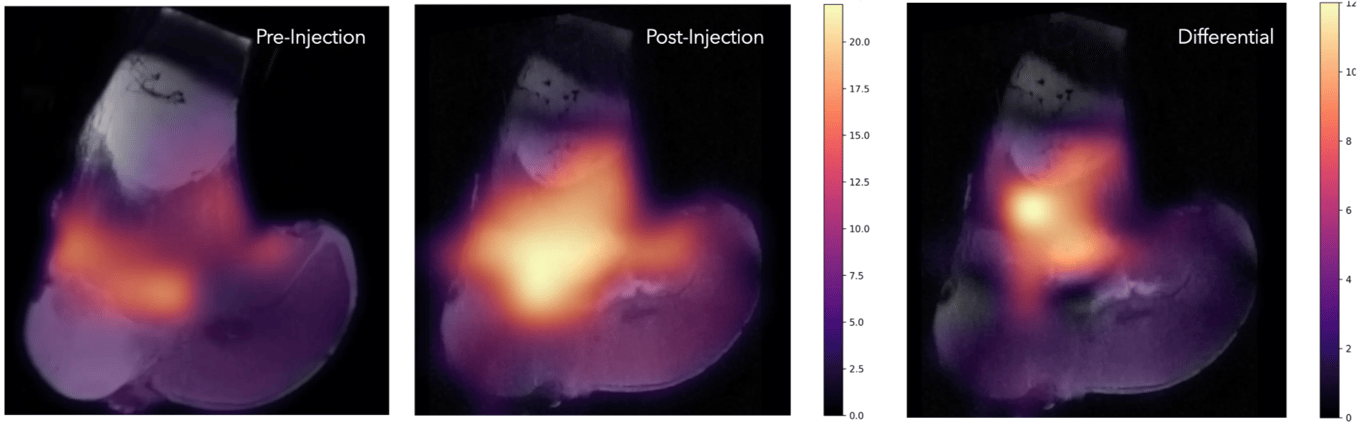
The MRI is positive for the pathological state of the knee, and so is Rilemo’s ERSI imaging. The fused images below show the specimen before injection (left, healthy), after injection (centre, pathological), and the differential between the two (right), illustrating the potential of the technique to monitor therapies over time through colour overlays on conventional anatomical images.
A faint presence of fluids is also visible in Rilemo’s colour overlay before the injection. This corresponds to the age of the bovine and to articulations still under growth, which retain a higher liquid content where the bone is still developing, an incidental finding that itself confirms the sensitivity of the technique to physiologically relevant fluid distributions.

What this validation proves
The device's colour-coded density maps are visually interpretable by radiologists on real biological tissues, enabling fast differentiation between normal and abnormal tissue states. Sensitivity to vascularised areas and fluids is sufficient to monitor physiologically relevant changes, opening future scenarios in paediatric diagnostics where MRI is challenging and ionising radiation should be avoided. Although this work uses a different anatomical target, it validates the underlying detection principle central to the device's imaging approach.
In-vitro phantom imaging (May 2024)
The in-vitro validation was conducted in a veterinary clinic with clinical staff involvement. This campaign demonstrated the feasibility of portable fluid imaging and validated the usability of the device's interface.
Methodology in brief
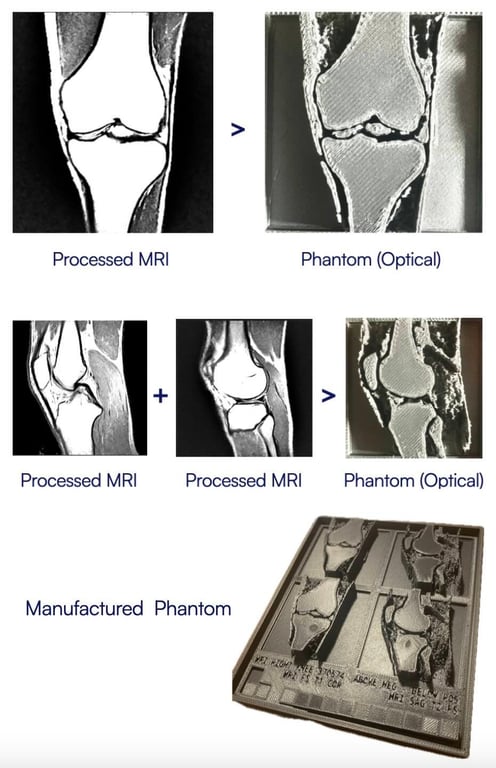
An MRI image of a 19-year-old male right knee was used to generate a flat synthetic phantom by combining multiple MRI slices in a 3D section. Two pairs of sections (one coronal and one sagittal) were 3D-printed in a variable mixture of ABS polymer and carbon fibre, chosen to approximate the dielectric properties of the tissues in the area of interest. One pair was printed solid, the other with superficial cavities into which edema-simulating fluid could be injected. The phantom was imaged with X-rays, with Rilemo’s prototype, and the results were superimposed.
Results
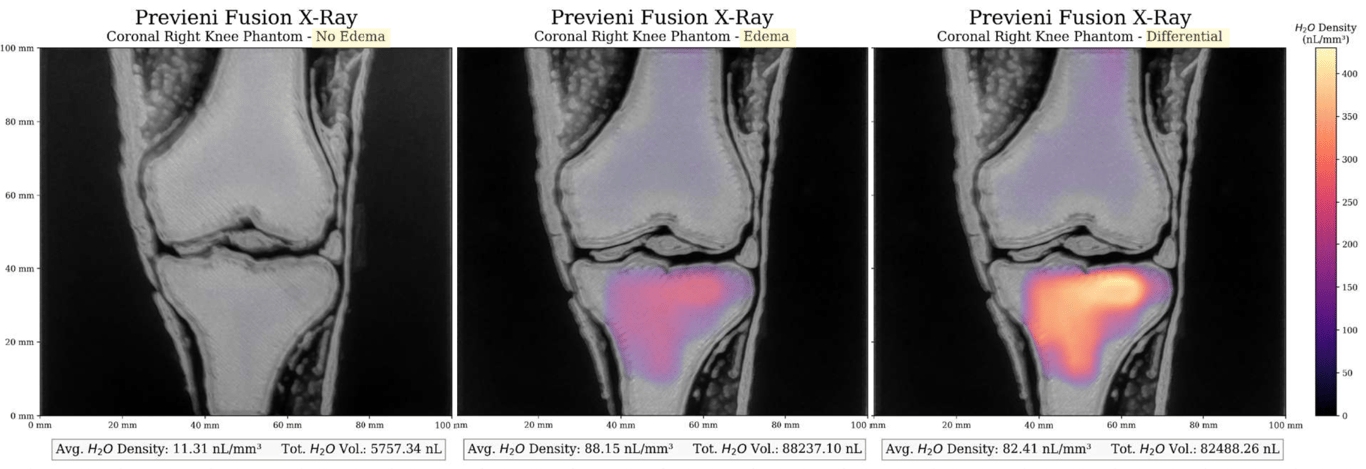
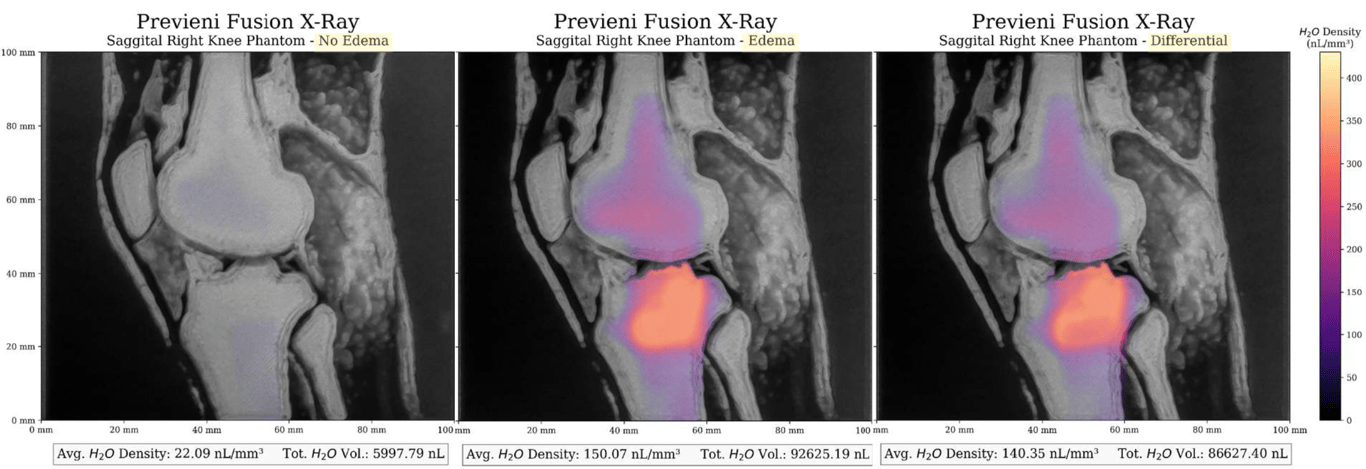
The fluid was injected at quantities as low as 90 µL, a volume estimated to be at least 20 times smaller than the smallest fluid pocket detectable by an MRI. X-rays were negative for any finding in both the healthy and the pathological case, while ERSI gave a strong positive finding in the pathological case, visualised through bright colour intensity on the differential image.

What this validation proves
The device detects fluid at sensitivity levels well below conventional MRI on phantom targets, with images that are clear and easy for clinicians to interpret. The differential imaging approach was described by users as critical for identifying and tracking pathological changes and for adjusting ongoing therapies.
What comes next
This pre-clinical portfolio is the basis on which Rilemo is entering its first-in-human clinical investigation, funded by this Seed round. The clinical investigation will be a non-inferiority study against CT for the defined acute neurological use case. A second cadaver experiment with the industrialised prototype is also planned during the pre-clinical phase, alongside continued refinement of the deep-learning, non-differential reconstruction pipeline. Full details are provided in the Roadmap section.
This Data Room is provided to you by:

Rilemo S.r.l.
Via Don Sebastiano Colleoni 20, Robbiate (LC), 23899, Italy
EU VAT: IT 04187140134 - ATECO: 26.60.02
PEC: - rilemo@legalmail.it
